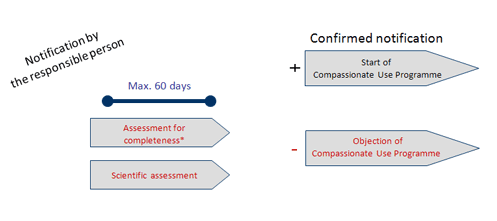
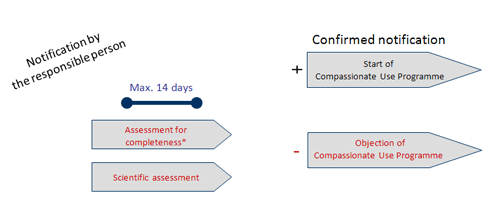
European Medicines Agency Domenico Scarlattilaan 6 1083 HS Amsterdam The Netherlands Subject: IV Zanamivir Procedure No. EMA/H

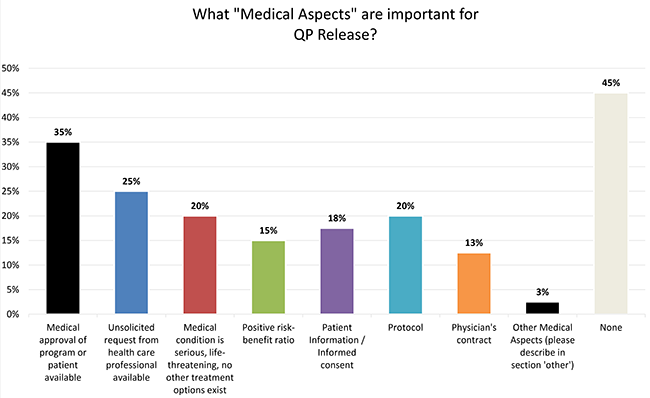
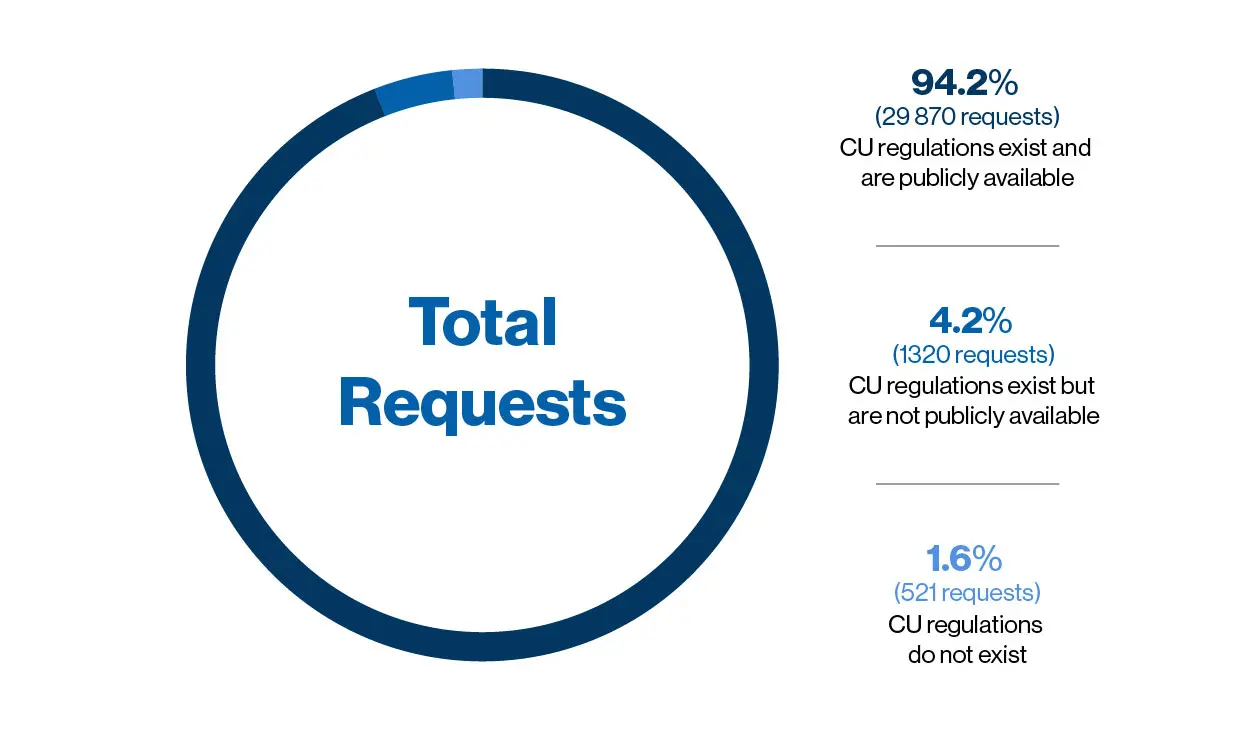
Manufacturer's Compassionate Use Policies: Companies with Posted Policies More Than Doubled Since September 2016
Compassionate Use Navigator Information for Physicians Application Process Overview Apply to a Drug Manufacturer

Compassionate use programs and the European regulatory system Filip Josephson M.D., Ph.D. Clinical Assessor. - ppt download