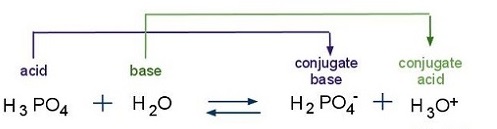
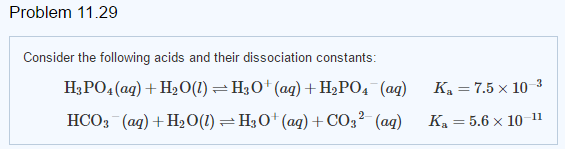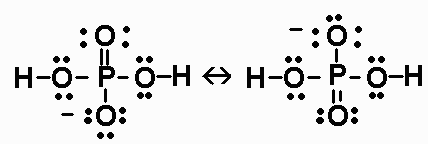SOLVED: For the following reaction; choose the correct answers from the drop down menu to complete the sentences: HC03 + HzPO4 2 + H3PO4 C03- Conjugate base of HCO3- is Conjugate acid

Acid-Base Test Review Definitions of Acids and Bases 1. Which of the following are Arrhenius acids? a. H2O b. H3PO4 c. NH3 d. H2SO3 2. Which of the. - ppt download

SOLVED: Compare the conjugate bases of these three acids Acid 1: carbonic acid H-COz Acid 2: phosphoric acid . HzPO4 Acid 3: hydrogen sulfite . HSO3 WVhat is the formula for the

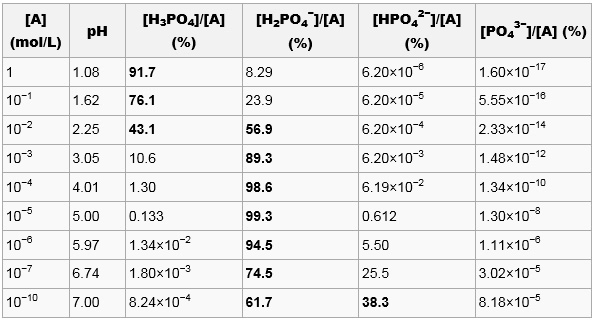
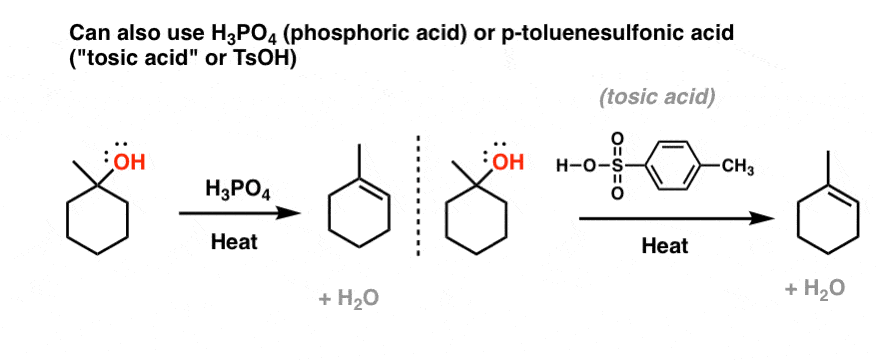
Buffer Calculations for Polyprotic Acids A polyprotic acid can form buffer solutions in presence of its conjugate base. For example, phosphoric acid can. - ppt download

15) Circle one and tell whya. CH3COOH is a n) (acid, base, salt)b. NH4Cl is a(n) (acid, base, salt)c. KOH - Brainly.com
![In the acid base titration [H3PO4(0.1 M)+NaOH(0.1 M)] e.m.f of the solution is measured by coupling this electrodes with suitable reference electrode.When alkali is added pH of solution is in acoodance with In the acid base titration [H3PO4(0.1 M)+NaOH(0.1 M)] e.m.f of the solution is measured by coupling this electrodes with suitable reference electrode.When alkali is added pH of solution is in acoodance with](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/32521666_web.png)
In the acid base titration [H3PO4(0.1 M)+NaOH(0.1 M)] e.m.f of the solution is measured by coupling this electrodes with suitable reference electrode.When alkali is added pH of solution is in acoodance with

Use your understanding of molecular structure to explain why the conjugate bases of acids like formic acid CHOOH, acetic acid CH3COOH, and phosphoric acid are only stable enough to be weak acids;
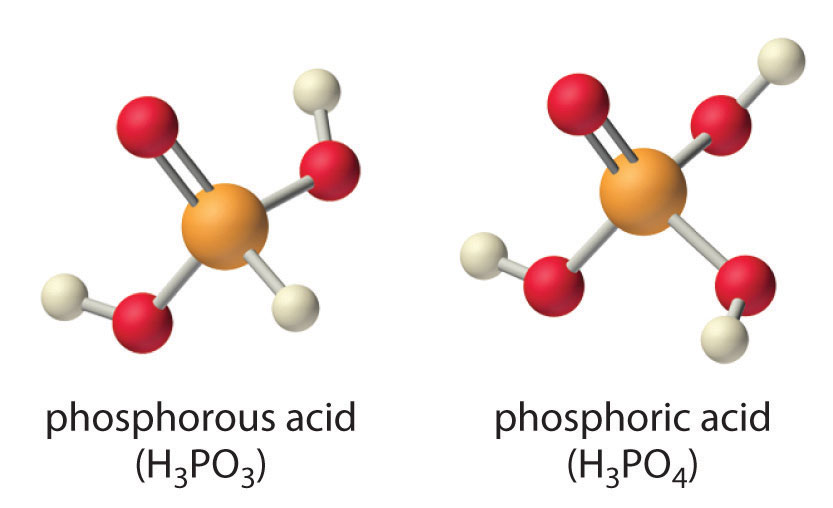
![Orthophosphoric Acid (H3PO4) [Phosphoric Acid] - Structure, Formula, Synthesis, Properties, Uses and FAQs of Orthophosphoric acid. Orthophosphoric Acid (H3PO4) [Phosphoric Acid] - Structure, Formula, Synthesis, Properties, Uses and FAQs of Orthophosphoric acid.](https://cdn1.byjus.com/wp-content/uploads/2018/11/phosphoric-acid-structure.png)