Acid and Base Equilibria Electrolytes Strong Conduct electricity Weak Poor conductors of electricity Nonelectrolytes Do not conduct electricity. - ppt download

SOLVED: 1. Answer and show all work: a. Are solutions of ammonium bromide, NH4Br; acidic, neutral or basic? Explain, using a chemical equation to demonstate your point. b. Explain hos sodium bicarbonate,

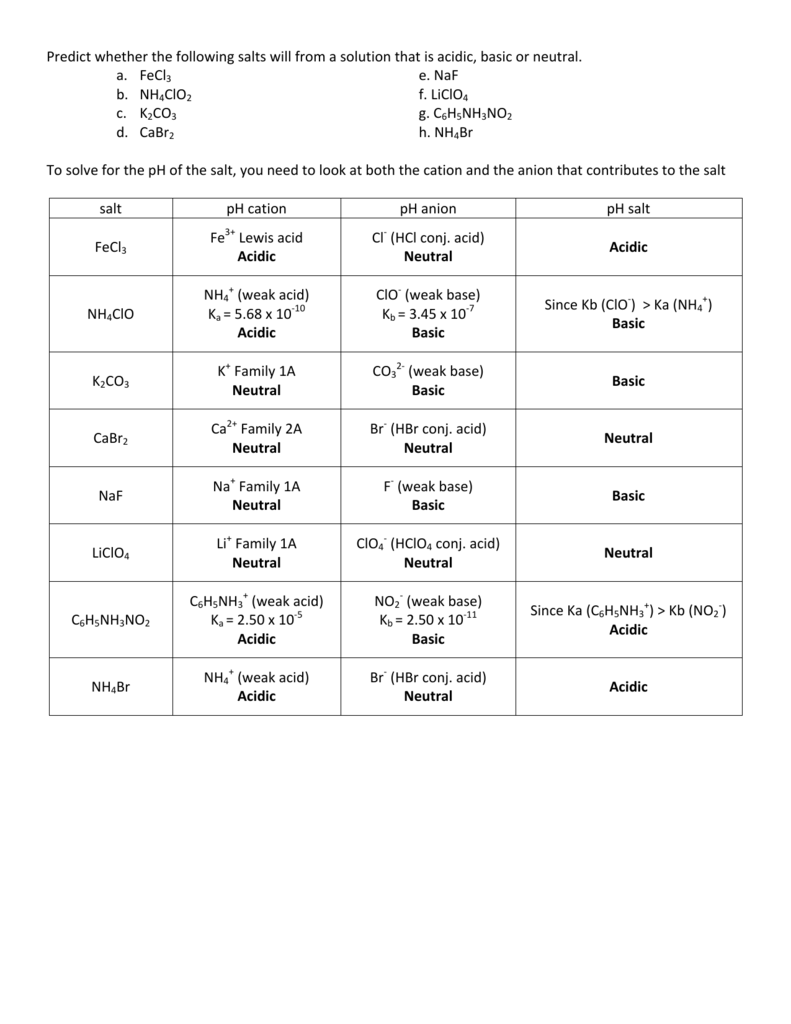
Chapt 12 Practice problems soln - Introductory Chemistry 030 (Prof. J. R. Tolman) Acids and Bases - Studocu
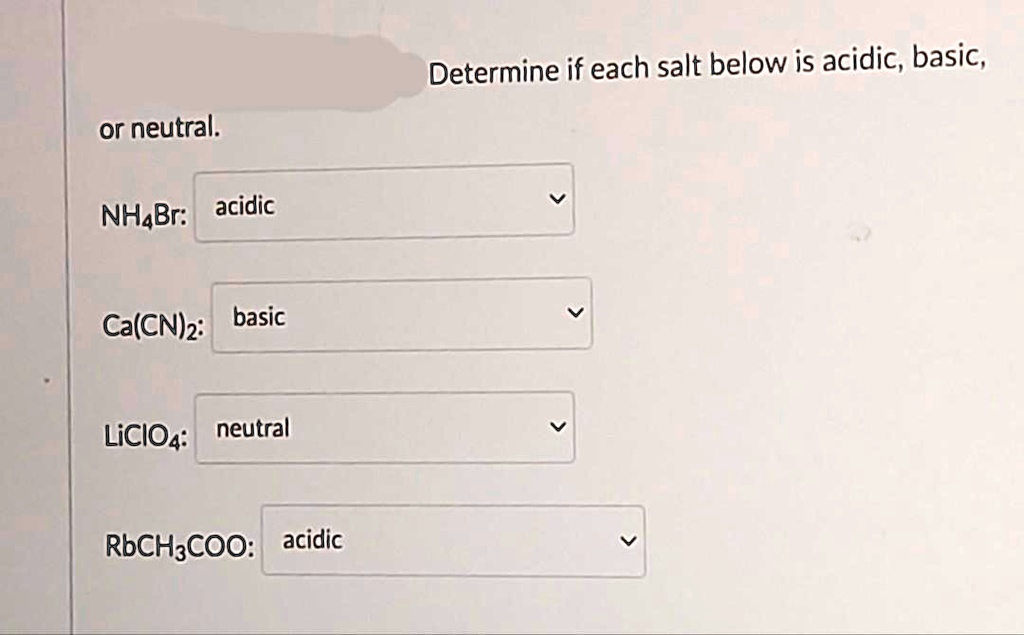
SOLVED: could you check if im right will rate thumbs up asap Determine if each salt below is acidic, basic or neutral. NH4Br: acidic Ca(CN)2: basic LiCIO4: neutral RbCH3COO:acidic

SOLVED: Predict whether aqueous solutions of the following compounds are acidic, basic, or neutral compound solution is .? acidic A(C1o4) , basic neutral acidic NH4Br basic neutral acidic K Br basic neutral