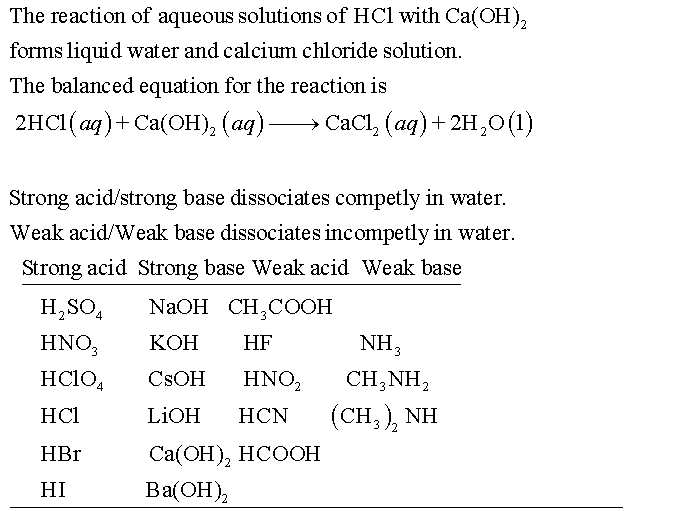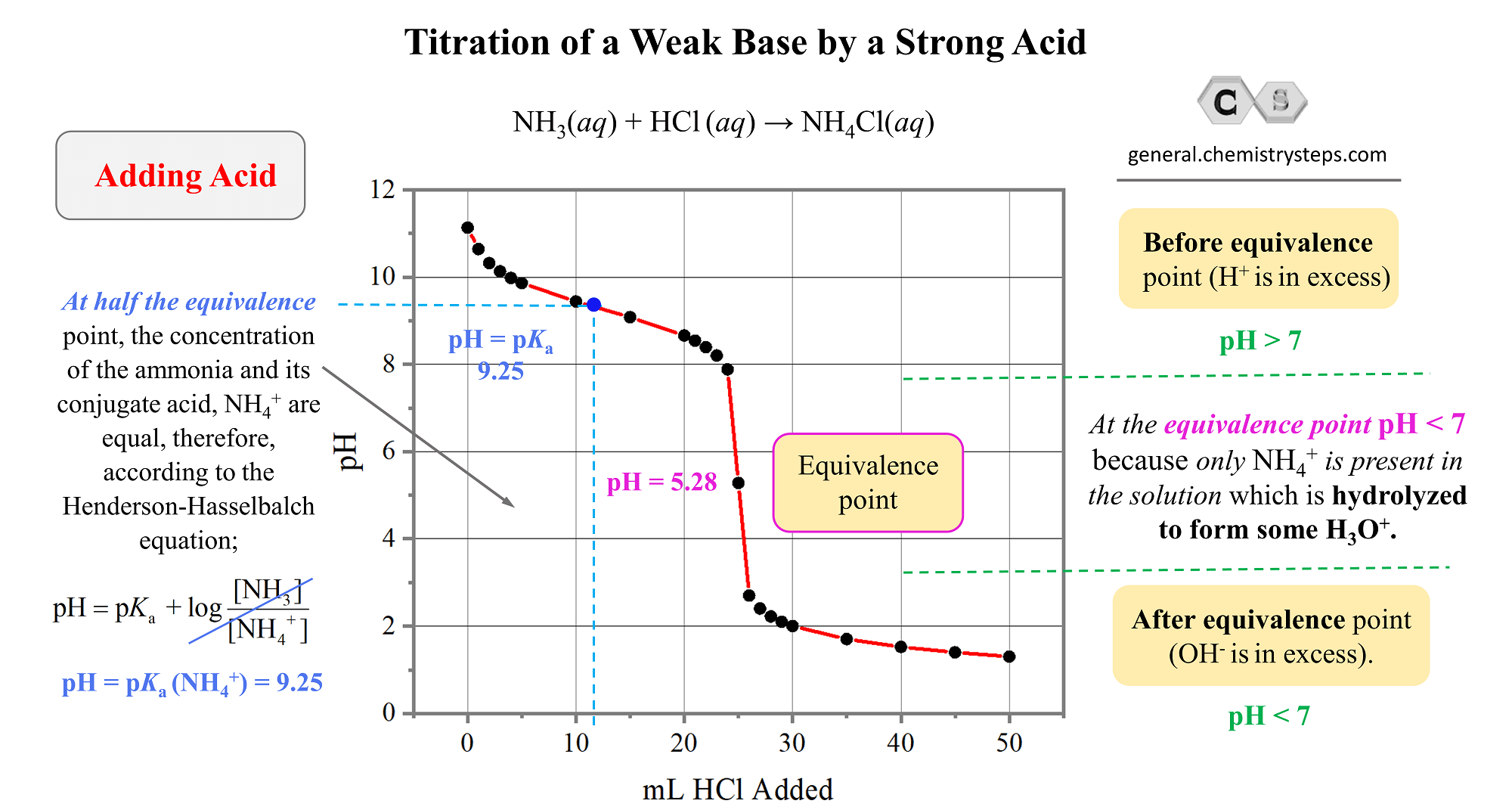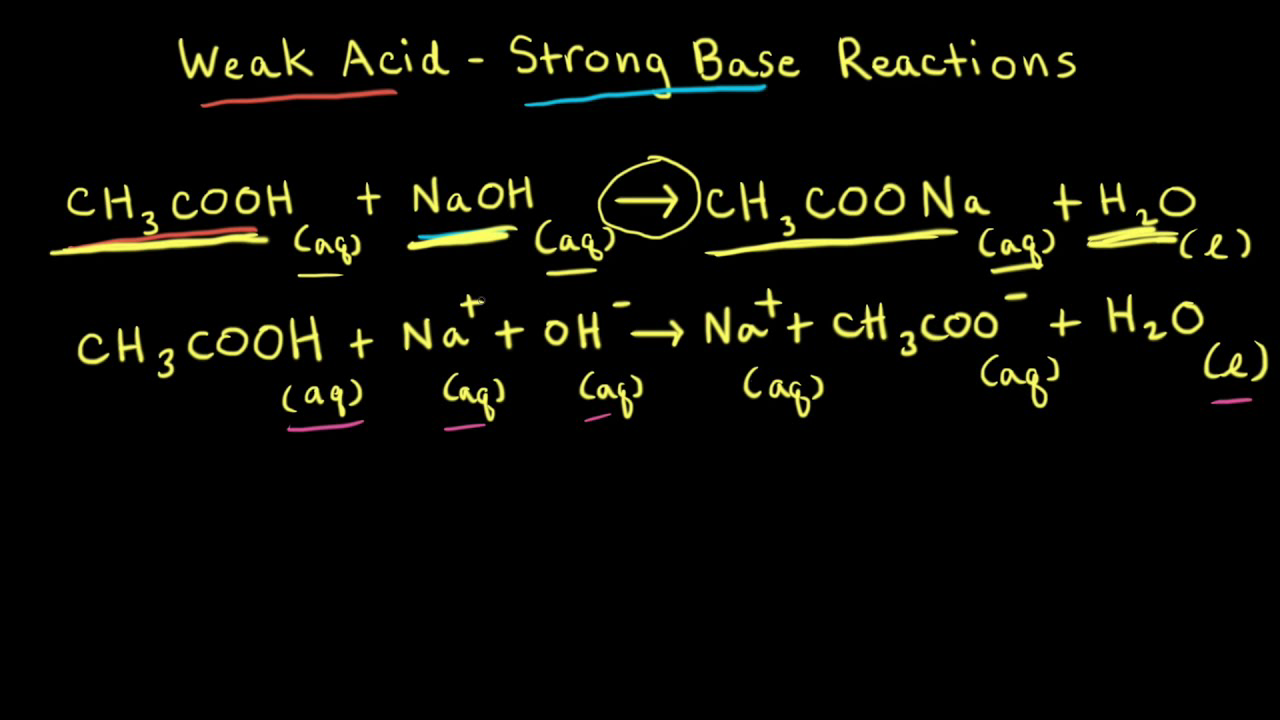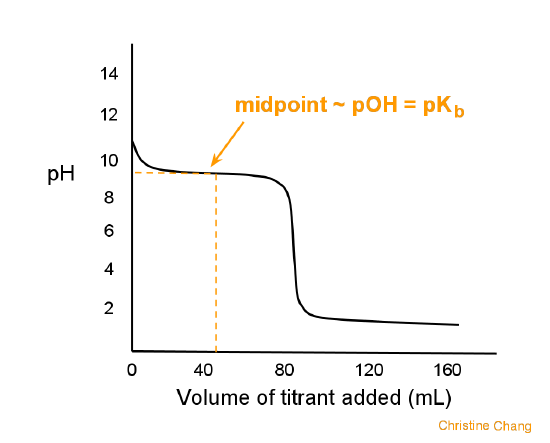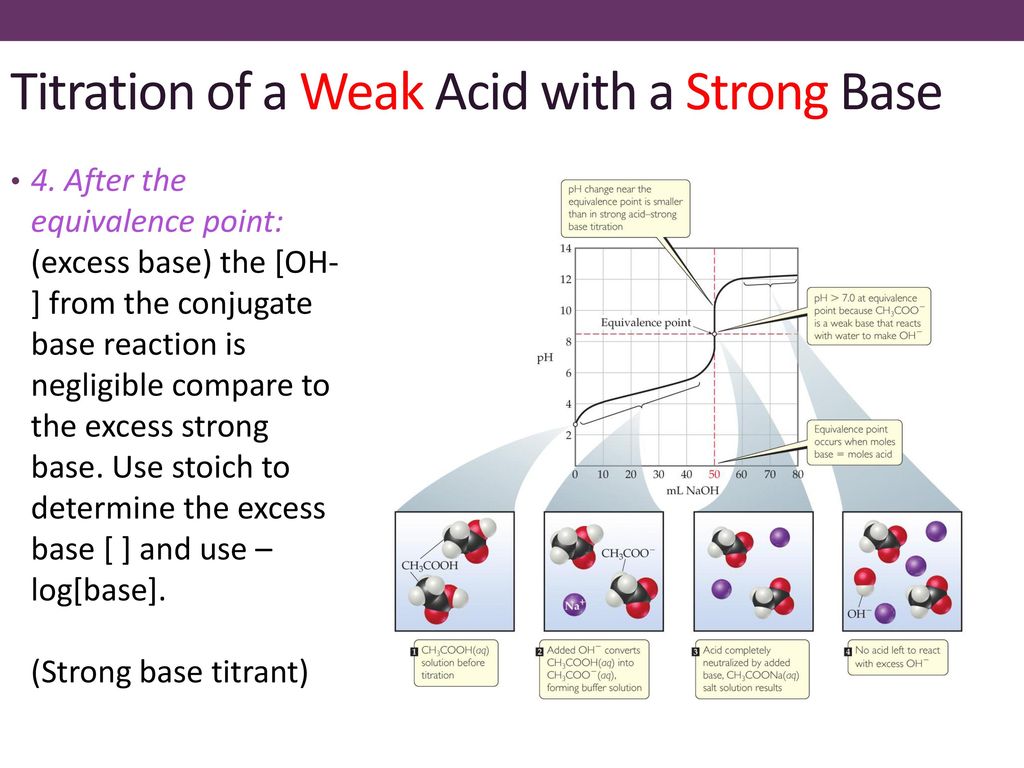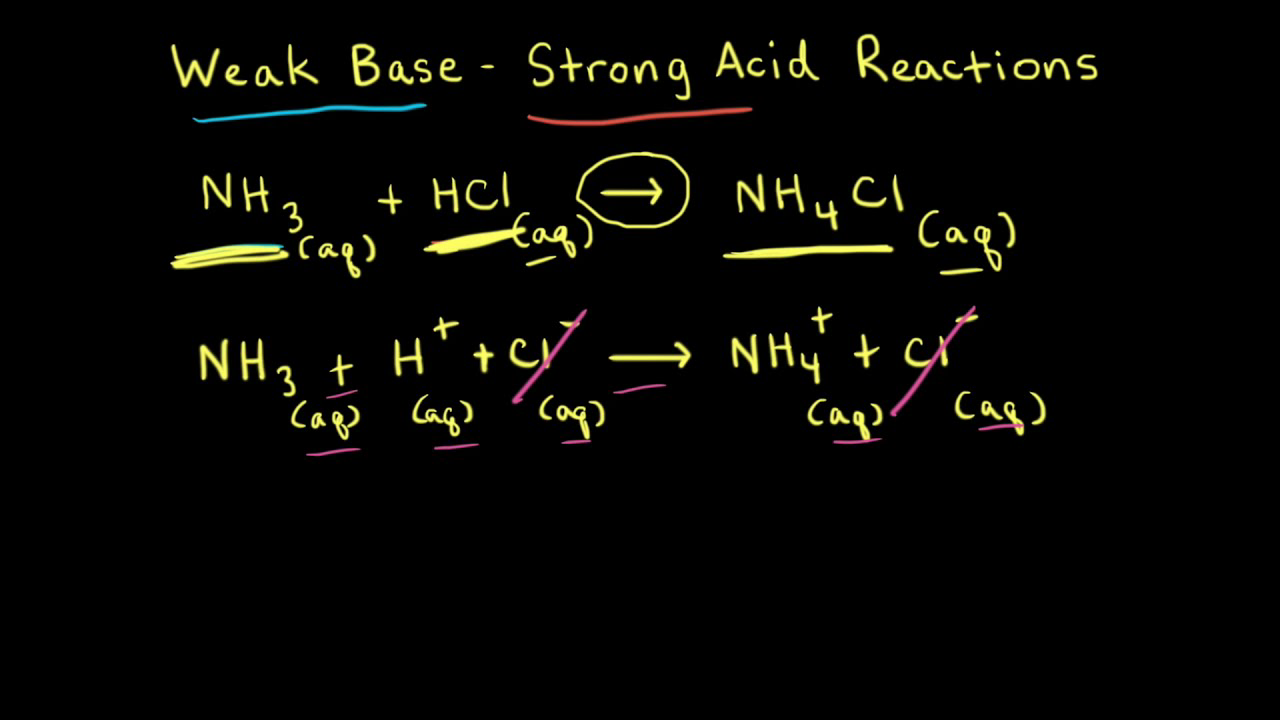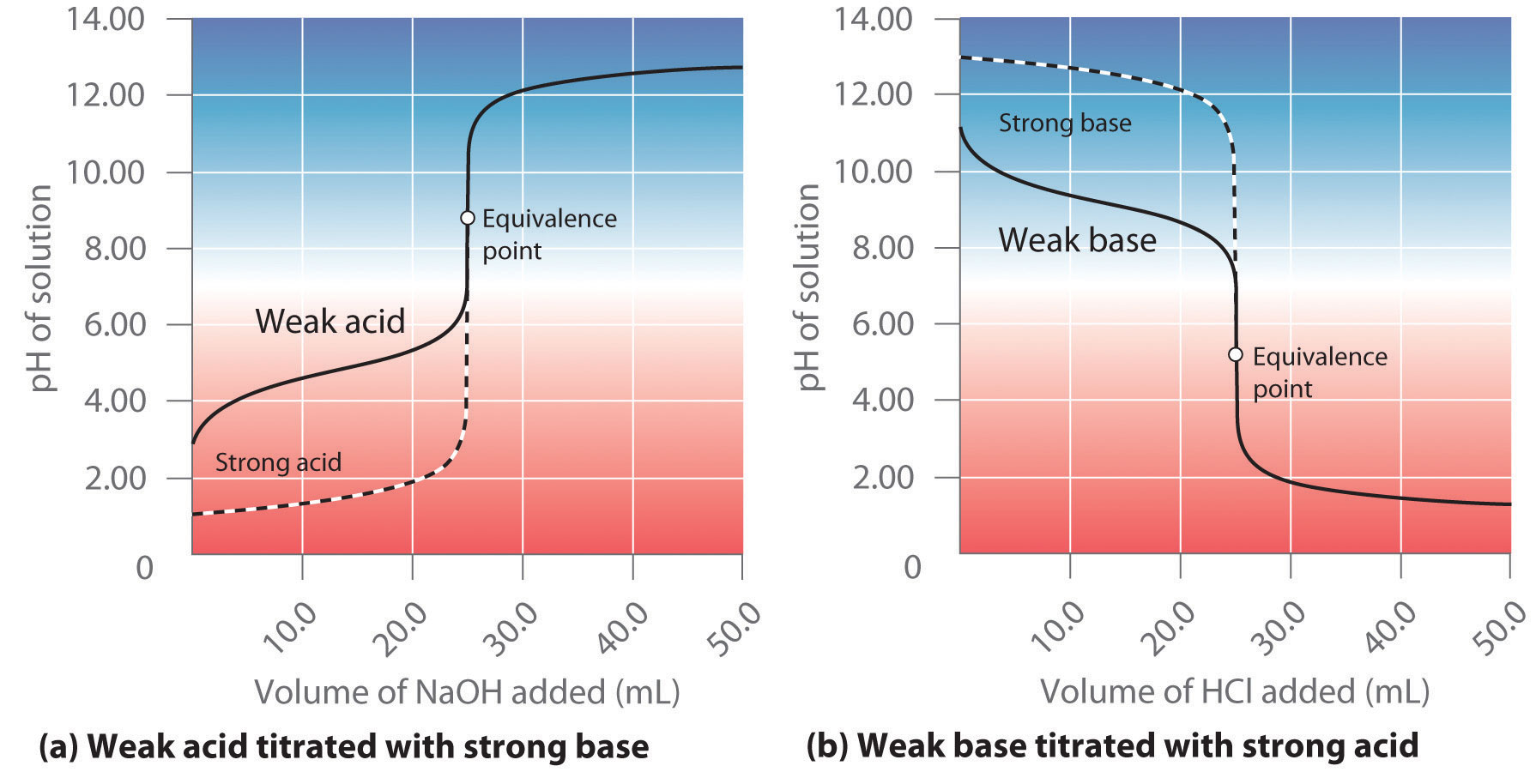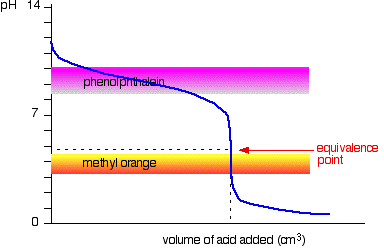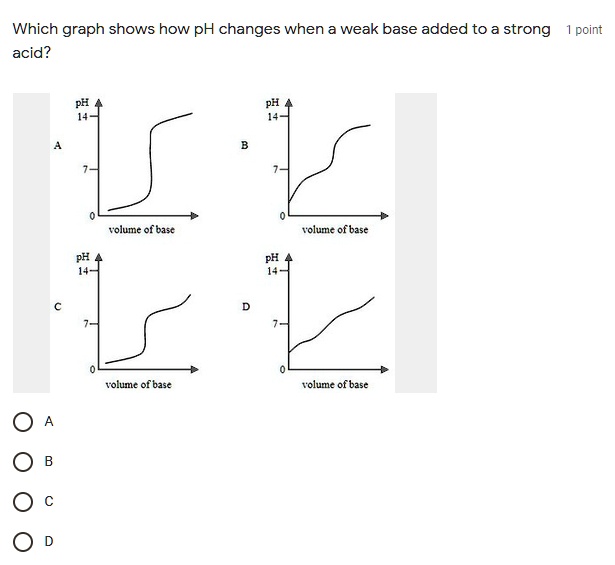
SOLVED: Which graph shows how pH changes when a weak base added to a strong acid? point rolumic ofbast Frclumc of basc rolumc of base rolumc of basc
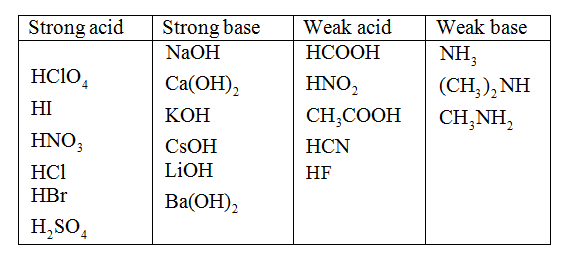
Classify each substance as a strong acid, strong base, weak acid, or weak base - Home Work Help - Learn CBSE Forum